Activity 1: Temperature & Heat
Examine the list of "Need to Know" words below. How do you think they relate to the Kinetic Molecular Theory? Before you start reading “Kinetic Molecular Theory Explained” below and watching the videos below. Brainstorm a list of 4 to 5 ideas about how the words listed below relate to the Kinetic Molecular Theory.
|
|
Your brainstorming should be recorded in your individual Google Document, Energy Transfer – Temperature, Thermal Energy, & Heat – Individual Thoughts. You are then to share your predictions by joining others in the class discussion forum, Energy Transfer – Temperature, Thermal Energy, & Heat – Activity 1.
Once you have finished your brainstorming you should move on to Time to Research portion of this activity. While you are reading the notes below and watching the videos, you should be making notes in your individual Google Document, Energy Transfer – Temperature, Thermal Energy, & Heat – Individual Thoughts. At the end of the activity you will be using these notes to create a concept map of the information you have learned.
Once you have finished your brainstorming you should move on to Time to Research portion of this activity. While you are reading the notes below and watching the videos, you should be making notes in your individual Google Document, Energy Transfer – Temperature, Thermal Energy, & Heat – Individual Thoughts. At the end of the activity you will be using these notes to create a concept map of the information you have learned.
Time to Research (Read, Watch, and Take Notes)
Kinetic Molecular Theory Explained
The kinetic molecular theory explains that particles in matter are in constant motion. Matter has thermal energy due to the kinetic and potential energies of its particles. Heat is the amount of thermal energy transferred from a warmer area to a cooler area. Heat transfer occurs by collision between particles (conduction), the movement of fluids (convection), or the movement of electromagnetic waves (radiation). (Sander, et al., 2008, p. 424).
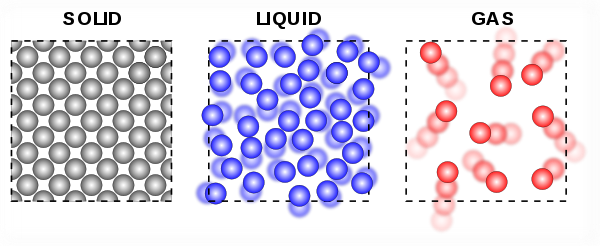
The kinetic molecular theory explains that all matter is made up of tiny particles.
The particles of a substance move differently for different states.
The kinetic molecular theory explains that particles in matter are in constant motion. Matter has thermal energy due to the kinetic and potential energies of its particles. Heat is the amount of thermal energy transferred from a warmer area to a cooler area. Heat transfer occurs by collision between particles (conduction), the movement of fluids (convection), or the movement of electromagnetic waves (radiation). (Sander, et al., 2008, p. 424).
The kinetic molecular theory explains that all matter is made up of tiny particles.
- These atoms and molecules are constantly in motion.
- Kinetic energy is energy due to motion.
The particles of a substance move differently for different states.
- In solids, particles vibrate slightly but do not change position.
- In liquids, particles vibrate more and move around within a set volume.
- In gases, particles vibrate greatly and move around to take all volume available.
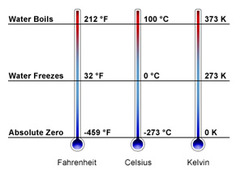
Temperature is the average of the kinetic energy of the particles of a substance.
- The movement of particles is what we measure as temperature.
- Temperature is measured using a Fahrenheit, Celsius and/or Kelvin scale.
Thermal energy is the total energy of all the particles in a substance.
Heat is the amount thermal energy that transfers from an area of higher temperature to an area of lower temperature.
- Thermal energy = all kinetic energy + all potential energy
- Kinetic energy is the energy of movement.
- Potential energy is the energy of position = how much the particles vibrate, and therefore how much space they take up.
Heat is the amount thermal energy that transfers from an area of higher temperature to an area of lower temperature.
Videos
|
Kinetic Molecular Theory fn Matter by Tutor Vista
|
- Misconceptions About Temperature by Veritasium
- Kinetic and Potential Energy by pheonixfilmandvideo
Check-In - To Be Completed Before Moving On
Let's have a check in. Create a Concept Map using the notes you recorded in your Google Document Energy Transfer – Temperature, Thermal Energy, & Heat – Individual Thoughts.
You can use any of the applications previously mentioned throughout the course (CMAP, Lucid Chart, or Simple Mind), you can use a new application, you can draw it out on a piece of paper and take a photograph of it to share. It is your choice of which technology to use to create the Concept Map.
After you have completed the Concept Map proceed to the class discussion forum, Energy Transfer – Temperature, Thermal Energy, & Heat – Activity 1 and share your Concept Map. You are then invited to look at concept maps made by your fellow students and to make thoughtful comments, generating further discussions on your learnings.
You can use any of the applications previously mentioned throughout the course (CMAP, Lucid Chart, or Simple Mind), you can use a new application, you can draw it out on a piece of paper and take a photograph of it to share. It is your choice of which technology to use to create the Concept Map.
After you have completed the Concept Map proceed to the class discussion forum, Energy Transfer – Temperature, Thermal Energy, & Heat – Activity 1 and share your Concept Map. You are then invited to look at concept maps made by your fellow students and to make thoughtful comments, generating further discussions on your learnings.